 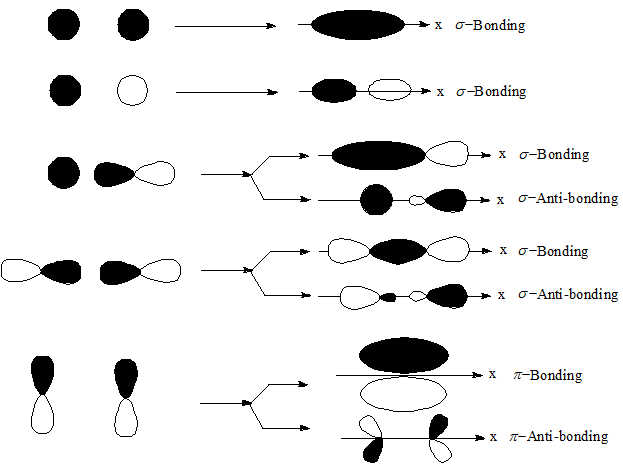
The orbitals would not be able to overlap, so the connection between the atoms would be lost.\) molecule. If one atom turns with respect to the other, the p orbital would have to stretch to maintain the connection. A s orbital is not affected when the atom at one end of the bond is rotated with respect to the other. True True/False: bonds formed from overlap of atomic s orbitals are always sigma bonds. A bond is a covalent bond in which the electron density is concentrated in the region along the internuclear axis that is, a line between the nuclei would. In other words, there are more nodes in the higher-energy orbitals than in the lower-energy ones.Īn important consequence of the spatial distribution or "shape" of a p orbital is that it is not symmetric with respect to the bond axis. True/False: A pi bond restricts rotation about the bond axis. The concept of sigma bonding is extended to describe bonding interactions involving overlap of a single lobe of one orbital with a single lobe of another. Just as the sigma-bonding orbitals display progressively shorter wavelengths along the bonding axis as they go to higher energy, so do the pi bonding orbitals. Sigma bonds are obtained by head-on overlapping of atomic orbitals. There will be both bonding and antibonding combinations. If two s orbitals directly overlap then the bond formed is linear between the two nuclear centres and is called a sigma bond. Show how the atomic orbitals overlap when a bond is formed. Sigma bonds are a result of the head-to-head overlapping of atomic orbitals whereas pi bonds are formed by the lateral overlap of two atomic orbitals. The two p orbitals orthogonal to the bond axis can engage in p bonding. C3h4 Valence ElectronsBelow is the electron dot structure for a Nitrogen molecule: In the. In a main group diatomic species like dinitrogen, one p orbital lying along the bond axis can engage in s bonding. A similar picture could be shown for the other set of p orbitals. The second picture shows the result of the constructive (or destructive) interference. True/False: End-To-End overlap of orbitals results in a bond with electron density above
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
